Mirrhia at A3P Congres
at booth O41!
We have all been impacted by these long periods of confinement and the absence of events and face-to-face meetings. Although we have found the solution with these tools that we certainly could not do without. And in a way it’s good as it allows us to keep in touch.
After ContaminExpo in Paris, we will be present in Biarritz for the 32nd edition of the famous A3P international congress. It is a must for all of us to be there.
For our Mirrhia team, participating in this congress is the perfect opportunity to discuss with you about your daily challenges, the issues your organization can encounter and which ultimately have a direct impact on the patient safety.
The A3P international congress will be held from 23 to 25 November at the Bellevue congress center in Biarritz.
On the program of this congress, the publication of the final version of the new Annex 1 will be at the center of the exchanges but also the new technologies, the remote audit (which has almost become the standard) without forgetting the exchanges between exhibitors on all the themes linked directly or indirectly to sterile matter.
The A3P international congress is also a series of workshops that will allow you to increase your theoretical or practical knowledge on one or the other topics thanks to the experts in technical or regulatory fields.

If you want to ask us a question or start a discussion with our Mirrhia experts, get in touch now.
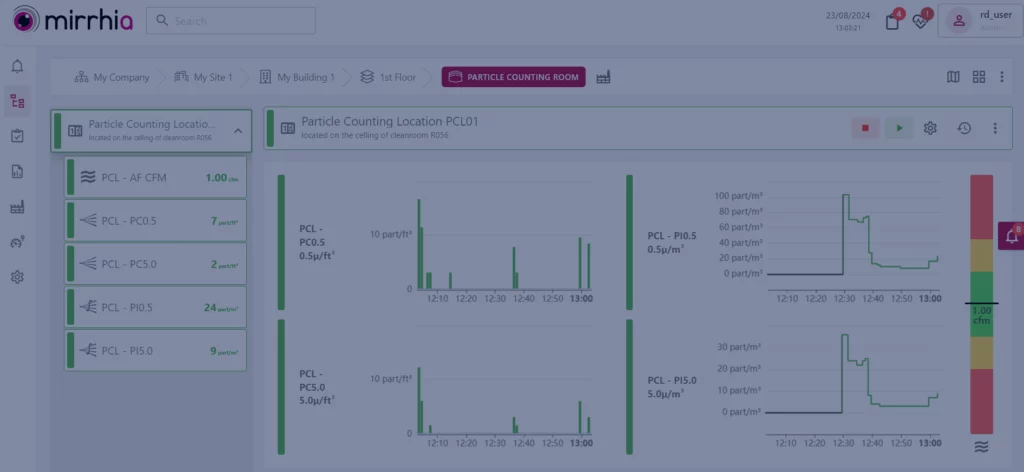
Mirrhia 2.5 is there!

Omega, Mirrhia’s Italian partner, in the spotlight in Genova Impresa

Arrival of Etienne Van den Bogaert as Managing Director
Mirrhia 2.4 is coming

Data Integrity in The Pharmaceutical Industry

Laborama 2024

Laboratory Temperature & Humidity Monitoring

Particle Counters | Case Studies & EMS in Pharma
